Consistency between value & price is in our access DNA
We are a healthcare consultancy conceiving and rolling out Global and French-setting applied Market Access and Pricing solutions for health technologies. We intervene at all lifecycle stages from earliest R&D stage to post-launch for defining and achieving rewarding market conditions.
Services
From earliest R&D stage to mature commercialization phase

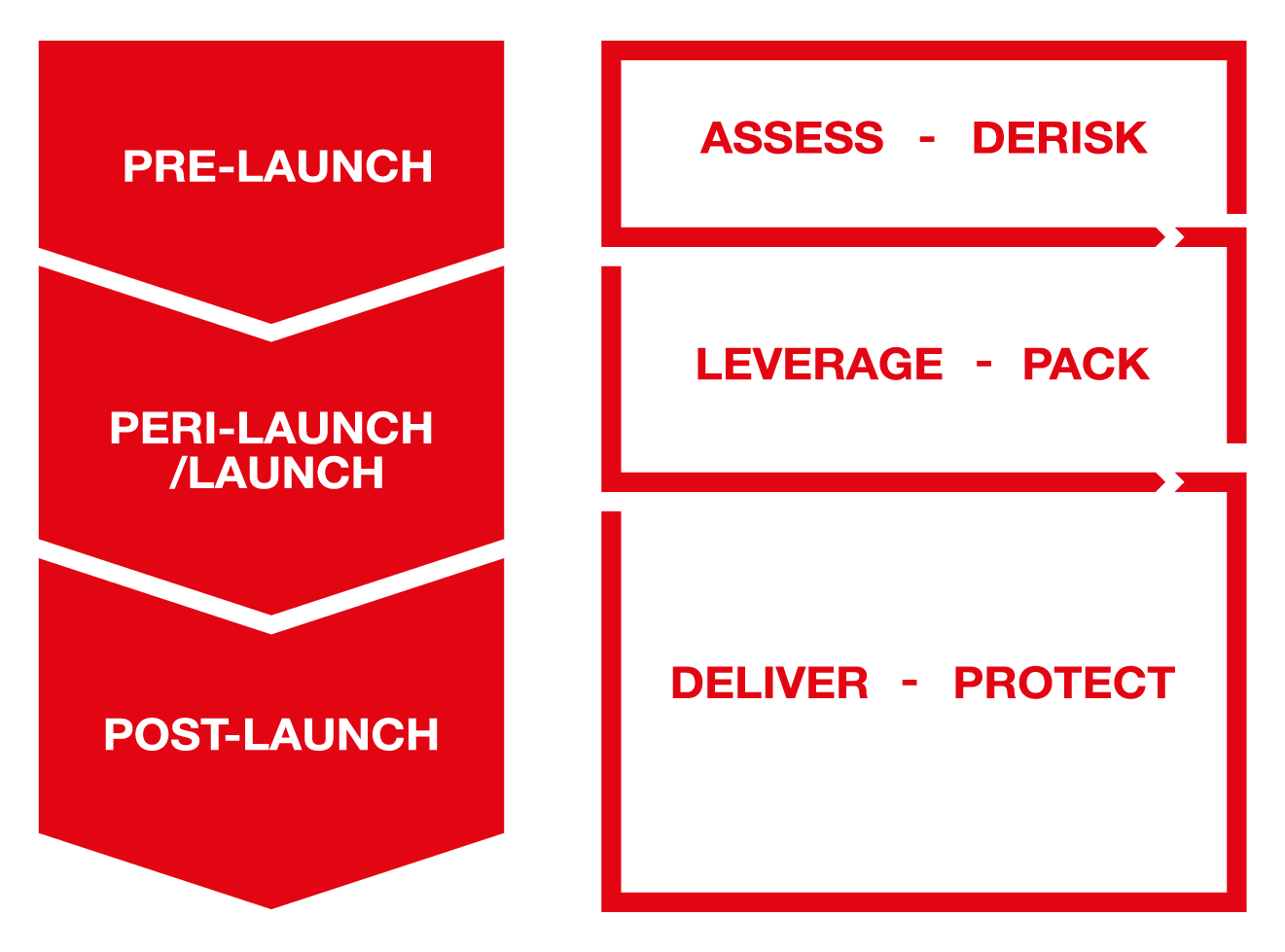
Strategic Access Road Map
Access Conditions and competitive landscape scanning, benchmarking
–
Target Product Profile definition
Asset profiling (endpoint selection, effectiveness extrapolation, biomarkers, patient subset…)
–
Value development plans
Clinical and economic value pillar identification, value de-risking Plan (incl. HEOR plan, ATC code..), Payer Evidence Set, Value Dossiers
–
Pricing
Price anchoring, Pricing strategies, new Pricing models
–
Early dialogues
With HTA bodies at EU & French levels
–
Interviews/Advisory Board
With Payers, proxy Payers & KoL
Early & Accelerated Access programs
–
Late dialogues
With HTA body at the EU level
–
HTA applications
At the French & EU levels
–
HTA body hearing
Preparation & attendance
–
Pricing
Policy definition, negotiation support & contracting insights
Post reimbursement commitment
Management
–
Real Life value
Confirmation
–
Drug reimbursement status
Renewal, re-evaluation
–
HTA application
For line extension
–
Pricing
Renegotiation, price increase, price cut minimization
R&D
Asset, pipeline, portfolio value assessement
-
Indication trade-off
-
Value audit of early stage programme
-
Clinician / Payer Advisory Board for entry into a new therapeutic area
MARKETING
Market Access strategy plan & roll out
-
Early Access & regular access support
-
Sales forecasting
-
Market Access project management

PARTNERING
Asset / portfolio assessment for licensing-in, -out & M&A exercices
-
Commercial Due diligence

MANAGEMENT
Corporate Market Access & pricing matters
-
Support to Public Affairs
-
Internal capabilities building /interim management / board membership

LEGAL MATTERS
Competition law exercises
-
Market Access litigations
-
Support in Patenting
About us
Dr. Véronique Foutel, the consultancy founder, is well acquainted with the scientific and economic evidence requirements of HTA evaluators & payers for both new assets and commercialized drugs and the way to generate this evidence in a timely manner.
Véronique gained this extensive drug market access and pricing experience through various job tenures in the French P&R Authorities, within the Pharma Division of Roche for 16 years, where she led the Global Strategic Pricing function, in the biotech field as Executive Committee Member of a listed late clinical stage biotech and now as strategic advisor to various biotech management teams and a finance boutique.
Speaker about the drug Market Access conditions in France and in Europe for les Echos Formation, Véronique gives master’s degree lectures at Paris Dauphine University.
Former hospital resident of Assistance Publique-Hôpitaux de Paris, Véronique holds a doctorate in Pharmaceutical Sciences from the University Paris V Descartes, and is a graduate of the Executive Health Economics programme of the Stockholm School of Economics and of the Roche Executive Leadership programme of the London Business School.
Partners
eponyme partners
Independent mid-cap mergers and acquisitions boutique website
Mc Arthur and Associates GmbH
Consultancy supporting drug discovery and early clinical development projects and large scientific alliances website
PharmaLex
Development consulting, regulatory affairs, quality management, compliance, pharmacovigilance, epidemiology & risk management worldwide website
Public Health Expertise
High-end life-sciences simulations, data analysis and market-access website
eXYSTAT
Biometrics & methodological expertise for clinical development activities carried out in pre-, peri- & post-launch website
Foxymed
Medical communication & consultancy website
LEAD-UP
Medical Communication & consultancy website
Our clients
Biotech


Pharma



Lifescience Stakeholders



Our events


Contact us

